On May 23, 2022, the Association for Accessible Medicines and its Biosimilars Council urged the Federal Trade Commission (FTC) to investigate the competitive impact of pharmacy benefit managers (PBMs) and their various abusive behaviors that undermine patient access to safe, affordable pharmaceutical care. In a letter responding to the FTC’s request for information “on the ways that large, vertically integrated PBMs are affecting drug affordability and access,” AAM outlines key PBM business practices that have a negative impact on America’s patients and the long-term sustainability of the nation’s competitive generics and biosimilars drug markets. Our comments were accompanied by a docket submission signed by patient, consumer, taxpayer, and other groups that bolstered AAM and the Council’s position. The diverse list of signatories includes National Consumers League, Allergy & Asthma Network, Global Healthy Living Foundation and Consumer Action, among many others.
Generics and biosimilars comprise 92% of prescription volume but only 16% of total drug spending. But the long-term sustainability of this market is threatened by excessive price deflation combined with harmful PBM business practices. And patients increasingly are harmed by PBM deployment of a distinct set of business practices that cause higher costs for and delayed access to generics and biosimilars.
PBM Practices Harm Patient Access to Lower-Cost Medicines
Two specific business practices are responsible for these harms. First, PBMs routinely place generics on formulary tiers with higher copays. Second, PBMs often favor inclusion of high-cost brand drugs, and the revenue opportunities associated with them, over lower list cost generics and biosimilars.
For instance, as a result of PBM tier placement decisions, yearly patient out of pocket spending on older generics more than doubled from 2010 to 2019 even as the price of those generics declined. In spite of the fact that generics and biosimilars represent the only segment of health care that consistently reduces costs, they represent almost two-thirds of all patient out of pocket drug spending. And more than half of Medicare beneficiaries were forced to pay twice for their generic – paying their Medicare premium as well as the full cost of the generic – due to the influence of PBM practices on plan behavior.
In addition, although new generics and biosimilars offer lower costs for patients, PBMs generate profit from high-dollar rebates and a range of fees on higher priced brand drugs and biologics. The well-documented formulary preference of PBMs for higher cost brand drugs can delay patient access to lower cost first generics or new biosimilar competitors. In fact, of the first generic drugs launched in 2020, only 66% were placed on formulary by commercial plans (less than 21% by Part D plans).
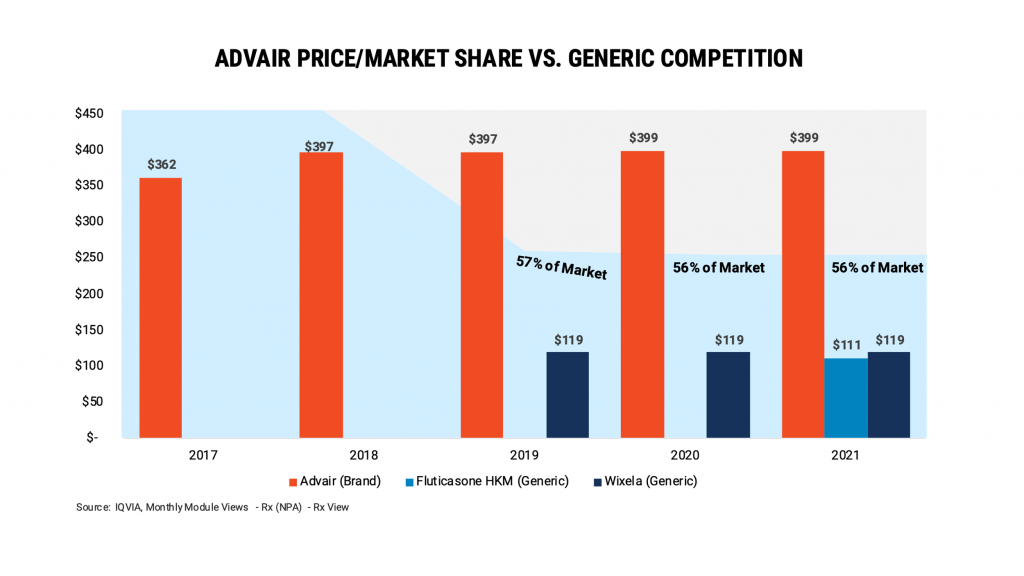
For instance, the COPD drug, Advair, has had generic competition for three years now at a 70% discount to the Advair list price. However, the brand product has managed to continue to maintain over 50% of the product’s market share. Such distortions cost payers money. And, most importantly, they cost patients.